Abdominal adhesions frequently occur after abdominal surgery. Stanford researchers prevented their formation in mice by blocking a molecular pathway.
Category: Surgery
Stanford surgeon repurposed her suturing skills to sew hundreds of masks
Stanford pediatric surgeon Janey Pratt converted her dining room to a factory, in order to produce cloth masks to protect people from COVID-19 transmission.
When your mother is a neurosurgeon: ‘It motivates me to push myself’
Reece and Alister Sharp, daughters of Stanford neurosurgeon Odette Harris, co-authored a children's book to share their experience.
Resuming surgery, other procedures safely in the era of COVID-19
In a podcast interview, a Stanford Health Care physician leader discusses how the system is keeping patients safe as surgeries and other procedures resume.
What it was like to get a pacemaker at 19, and how it changed my life: Part 2
College student Bea White writes about her pacemaker-implant surgery, and how her life has changed since having the procedure.
When I was a teenager, I found out I have a serious heart condition: Part 1
At 19, Bea White learned she needed a pacemaker to speed up her heart, which beat too slowly because of a condition called heart block.
Behind the scenes with a Stanford pediatric surgeon
A day in the life of Stephanie Chao, a Stanford pediatric surgeon, researcher and mother trying to live in the moment and balance the chaos.
More prep before surgery, less pain after
Stanford physicians have developed ways to better prepare patients physically and mentally for surgery, helping them to feel less pain during recovery.
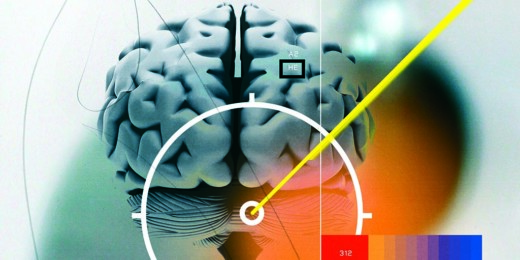
Brain surgery advances — precision targeting
Lasers, heat maps, fluorescence and real-time imaging help guide surgeons who are developing new ways to enhance precision brain surgery.
The type of eye surgery may depend, in part, on the day of the week
Stanford scientists found patterns in how ophthalmologists chose to repair retinal detachments, based on days of the week.
Stanford surgeons innovate new biological breast implants
Surgeon Irene Wapnir and her colleagues developed a new technique for creating biological breast implants for women who have undergone a mastectomy.
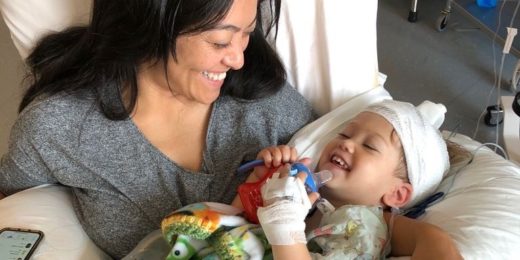
Two-year-old becomes youngest patient to have giant brain tumor removed through nasal cavity
A first of its kind surgery removed a problematic tumor from the brain of two-year-old Ari Ellman, allowing him to return to his life as a busy preschooler.
One bicycle crash and six years of recovery: Part II
After a bike crash, Anthony Macchio-Young has emergency neurosurgery at Stanford. In the conclusion of this two-part series, he shares how he is doing now.
One bicycle crash and six years of recovery: Part I
After a bike crash, Anthony Macchio-Young undergoes emergency neurosurgery at Stanford. But that's only the beginning of his journey to recovery.
A spray-on gel may help prevent internal scar tissue
Stanford researchers discover a gel that, when applied to animal hearts, vastly reduces the formation of adhesions, scar tissue that cause complications.
Helping young cleft palate patient feel like a star
10-year-old Mathias Dizon fulfilled a promise to sing the national anthem at the Stanford Children’s Health Cleft and Craniofacial Center's annual patient and family picnic.