Stanford scientists have taken important steps toward figuring out how to use immune therapy for a group of severe pediatric brain tumors known as atypical teratoid/rhabdoid tumors.
A new study, published recently in Nature Medicine, identifies a molecular target that enables engineered, cancer-fighting immune cells to recognize and attack the tumors while leaving healthy brain tissue alone. Using mouse models, the researchers also showed that giving the cancer-fighting cells directly into the brain was safer and more effective than administering the cells in a vein. The discovery will help the scientists plan upcoming human clinical trials.
"Clinical progress occurs stepwise: We have to come up with a treatment idea, then tweak, iterate and optimize it," said senior study author Crystal Mackall, MD. Mackall is a longtime leader in the development of chimeric antigen receptor T-cells, or CAR-T cells, for cancer treatment. CAR-T cells are immune cells engineered to recognize specific markers found on cancer cells and attack them. CAR-T cells form the basis of recently-introduced cancer immunotherapies, such as tisagenlecleucel (brand name Kymriah), which is used to treat leukemia and lymphoma.
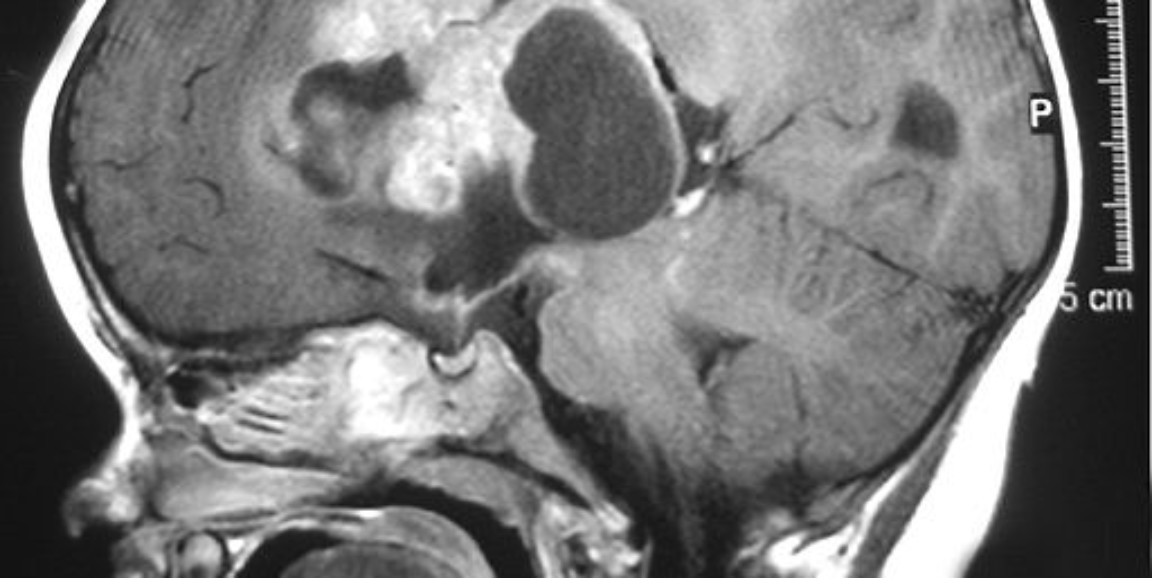
Experts want to expand the use of CAR-T cells to other cancers, including pediatric brain tumors. Surgery, chemotherapy and especially radiation treatments can have devastating side effects on a child's brain, so the idea of eliminating brain tumors with highly-targeted immune cells is very appealing. The tumors studied in the new research, atypical teratoid/rhabdoid tumors, are among the worst types of childhood brain tumors. They occur in babies and toddlers; and, with current treatments, patients with these tumors have a median survival time of only 17 months.
But scientists face two big challenges in using CAR-T cells for these tumors: In general, pediatric brain tumors don't have very many mutations, so they appear molecularly similar to healthy brain cells. And giving immune cells into the brain comes with its own safety challenges.
The new study addressed both problems. On the targeting front, it showed that a protein marker called B7-H3 is abundant on ATRT tumors but not present on healthy brain cells in babies and children. (The marker is normally expressed on fetal brain cells, but quickly disappears from healthy brain cells after birth.) When the scientists tested CAR-T cells that recognize and attack B7-H3, they found that the CAR-T cells were very good at getting rid of ATRT tumors in mice.
"Our most exciting finding was how effective the CAR-T cells were," said Johanna Theruvath, MD, the study's lead author. "They were extremely effective at eliminating all the tumors. We hope this finding will translate into people."
On the safety front, scientists are concerned about using CAR-T cells to treat brain tumors because immunotherapy can cause the release of large quantities of immune molecules called cytokines. Too many cytokines, in turn, can produce a lot of swelling, which is dangerous for the brain.
To avoid this problem, the researchers tried three routes of administering the CAR-T cells: They gave cells directly into the brain tumor; into the cerebrospinal fluid that bathes the brain; or into a vein. Administering cells to the cerebrospinal fluid worked best, Mackall explained.
"One of our big findings is that if we want to get the T cell to the brain tumor, we should just put it in the brain," she said. "It's kind of simple, but it was remarkable to see how powerful that simple maneuver is in terms of making the therapy more effective. You need about 10 times fewer CAR-T cells to cure the tumor than if you give the cells via a vein; and with fewer cells, there was less toxicity."
Cytokine levels were lower -- and safer -- in animals that received CAR-T cells in their cerebrospinal fluid than in animals that got the cells in a vein.
The researchers also found that CAR-T cells given directly into the brain persisted in the brain after the cancer was gone, and that some of these cells migrated out of the brain and stayed in other parts of the body.
"This is illustrating how little we know about the way these immune cells traffic in and out of the brain," Mackall said.
The persistent CAR-T cells were able to protect mice against more tumor cells introduced later, suggesting that the cells might offer long-term protection against cancer relapse. The researchers hope to offer a clinical trial using the CAR-T cells for adults with the brain tumor glioblastoma early next year. If that succeeds, they will work toward offering clinical trials for children.
Image by Marvin 101