Back in the fall of 2020, even before the Stanford pharmacy team learned which COVID-19 vaccines would hit the market first, they were already preparing for greatest impact.
"In pharmacy, our comfort zone is precision," said Deepak Sisodiya, PharmD, MHA, the Administrative Director of Pharmacy Services at Stanford Health Care. "That's the way we're trained, to be absolutely sure and 100% accurate. But in a pandemic, that's very tough to do."
With information about the virus evolving by the day, the pharmacy team had to constantly prepare for the unexpected. In the early days of the pandemic, that meant clearing out entire rooms and shoring up medication supplies for a possible surge in COVID-19 patients. By November, it meant scrambling to find the right kind of freezers to store vaccine.
Not your typical vaccines
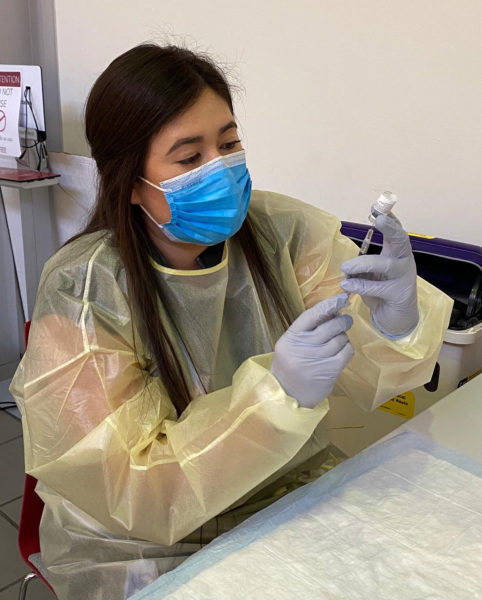
"Pfizer, Moderna; these are not your typical vaccines," said Brooke Betts, PharmD, a pharmacy administration resident, who played an integral role in managing vaccine flow. "There are a lot of very specific requirements in terms of storage and handling, as well as distribution."
The Pfizer vaccine, for instance, must be shipped overnight on dry ice and then stored immediately at -80°C in an ultra-low temperature freezer.
"Our supply chain team took immediate action and were preordering these ultra-low temperature freezers," Betts said. But of course, every other hospital in the country was preordering them too. "It was pretty wild, but we were working very hard, and we got lucky," Sisodiya said, eventually securing procurement for two of the freezers and borrowing one from the School of Medicine in the meantime.
But freezers were far from the only challenge. Because mRNA vaccines remain stable only at below-zero temperatures, the pharmacy team had to carefully coordinate how they would receive vaccine shipments, unpack the boxes, account for all the doses, and get the trays into the freezer -- all within minutes, before the temperature became too high.
"I'm not kidding, it's almost like a pit crew in NASCAR or F1 racing," Sisodiya said, with up to four pharmacy team members working in tandem. "It takes careful orchestration to do that in the right manner."
No dose wasted
Once defrosted, the Pfizer vaccine must be administered within five days. Especially early in the roll-out, with more than 10,000 Stanford Medicine employees awaiting vaccination and demand vastly outstripping supply, wasting even a single dose was unacceptable.
"That's where my role came in," Betts said, "in terms of making sure we had enough defrosted for patients, but not so much that we would waste it."
Soon after vaccinations began, the pharmacy team made an exciting discovery: both Pfizer and Moderna vials include enough vaccine to administer one or sometimes even two doses beyond what's on the label. Called "overfill," this extra vaccine provides a buffer in case some gets stuck in the bottle or syringe. But in this case, even before the drug companies gave the regulatory green light to use the extra doses, no one wanted to waste a drop.
"We were going to pull that sixth dose out regardless," Sisodiya said. "To get as many shots in arms as possible."
But accessing that overfill requires expert-level aseptic technique, and initially a pharmacist needed to be directly involved with drawing up each dose. Preventing waste also meant long hours: "We would have the vaccine center run until almost 11 p.m.," Sisodiya said. "If there was even a single dose left, we would roam the halls to find someone."
And because of the many levels of vaccine oversight, the pharmacy team would be up again by 4 or 5 a.m., preparing reports for the California Department of Public Health and the Centers for Disease Control and Prevention.
A teamwide effort
All across the pharmacy team -- and across Stanford Medicine as a whole -- employees stepped up to help. "Whether they were from inpatient, the Emergency Department, or Central Pharmacy," Betts said, "everyone was asking how they could help, when they could help next and how they could help more."
To date, more than 100 pharmacy team members have participated in the vaccine effort. "This took the whole village of pharmacy," Sisodiya said. "I can't thank my team enough, for their bravery and the way they rose to this cause."
Top photo, courtesy of Brooke Betts, shows the Pfizer COVID-19 vaccine being received at Stanford by pharmacy team members (left to right) Maria Deguzman, Daniel Fong and Adam Silva.




