In December 2014, Lasker Award-winning Stanford biochemist and structural biologist Jim Spudich, PhD, had a dream that changed the course of his research and led to a new understanding of -- and perhaps a breakthrough treatment for -- hypertrophic cardiomyopathy, a mostly inherited heart defect that affects 1 in every 500 people throughout the world.
Hypertrophic cardiomyopathy's defining clinical symptom is a hypercontractile heart. "It's as if you're out for a short run," Spudich says. "The problem is, you're doing that 24 hours a day, every day of your life." The upshot can be sudden death at an early age.
As I wrote in a recent feature article about this fascinating (and, remarkably, true) story:
The heart beats because it's made of muscle cells that rhythmically shrink in sync and then relax, pumping blood throughout the vascular system. This nonstop throbbing owes its rhythm to a pair of proteins that make all muscular contraction possible.
Hypertrophic cardiomyopathy can arise due to hundreds of different genetic mutations, most of them occurring in just a handful of proteins comprising the heart's muscle-contraction machinery. One protein in particular, myosin, has been on Spudich's mind since 1969 -- his unraveling of this contractile molecule's workings is what won him the coveted Lasker in 2012. Another key protein in this machine is called actin.
Here's a riddle that stymied Spudich for years: Usually mutations cause a protein not to work as well as the unmutated version does. But the mutations responsible for hypertrophic cardiomyopathy do the opposite! They somehow cause the protein to work 'better.' The result is a heartbeat that's too powerful.
The lion's share of those mutations affect myosin. A myosin molecule's general structure resembles a two-headed monster: two large globular "heads" protruding from a stalk-like tail. (See illustration below.) It's long been known that myosin sometimes adopts a posture in which its heads fold over and snuggle up against its tail, reminiscent of a sleeping flamingo with its head tucked under a wing.
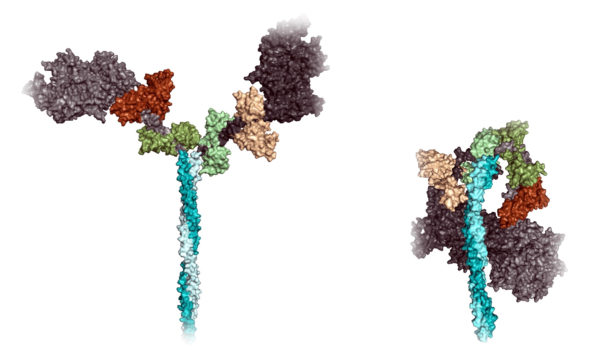
But nobody cared. To see why they should have, it helps to know how healthy muscles contract. From my article:
A cardiac muscle cell contains perhaps a hundred repeating structural subunits called sarcomeres, [each] composed of myosin-rich 'thick filaments' alternating with parallel 'thin filaments' made of actin...
In response to electrical impulses traversing the heart, the myosin heads protruding from the thick filament chomp down on the nearest actin filament, then tug against it like sailors tugging in tandem on a rope, pulling the sarcomere walls closer together and making the muscle fiber contract, before relaxing again. That's a heartbeat.
And if those myosin sailors are tugging too hard on those actin ropes? That's hypercontractility, the hallmark of HCM.
But why would this happen? Spudich and his labmates had been focusing on this question for years. And they'd been stumped.
One night as Spudich lay awake in bed late, wondering what clue he'd been overlooking, his wife told him to take a break. She slipped him some bedtime reading, a murder mystery. Spudich knocked off a few chapters before falling asleep, only to awaken in the dark with the germ of a vision that would solve the mystery he'd been brooding over for more than a year.
I think I'll stop here and leave you in suspense. Want to know more? Read the article. But I will leave you with this: Amazing discoveries and inventions, from the sewing machine to the periodic table and the theory of relativity, have sprung from the dream of sleeping scientists.
Photo by Edward Caldwell; Protein image courtesy of Jim Spudich




