In my part one on the bare-bones basics of viruses, I described how your average virus -- an essentially inert particle on its own -- manages to enter cells, hijack their molecular machinery, make copies of itself and move on out to infect again.
That just scratches the surface. Of the millions of different viral species identified so far, only about 5,000 have been characterized in detail. Viruses come in many shapes and sizes -- although they're all small -- and infect everything, including plants and bacteria. None of them work in precisely the same way.
With crucial assistance from Stanford virologist Jan Carette, PhD, here's more information about some viruses worth knowing a bit about.
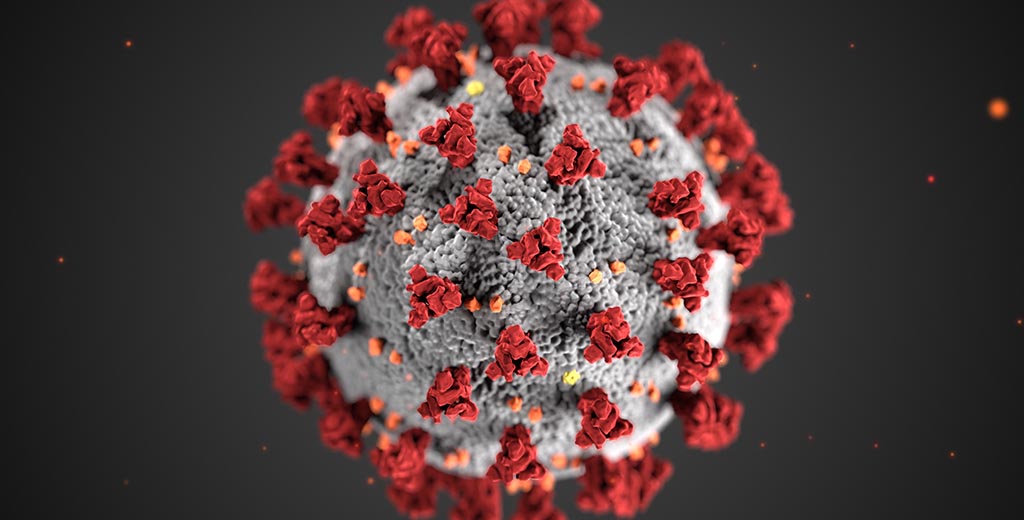
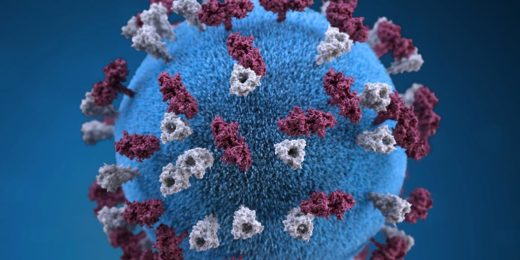
Coronavirus, for example.
Enveloped viruses generally tend to be less hardy when they're outside of cells because their envelopes are highly vulnerable to degradation by heat, humidity and the ultraviolet component of sunlight.
This should be good news when it comes to coronaviruses, which are enveloped viruses. However, the bad news is that the coronavirus can be quite stable outside of cells because spike-like proteins protruding from its envelope shield it from direct contact, enabling it to survive on surfaces for relatively long periods. (Still, soap or alcohol-based hand sanitizers do a good job of disabling it.)
As mentioned in part one, viruses use proteins that are sitting on cells' surfaces as docking stations. These cell-surface proteins aren't there to welcome viruses aboard -- they've got day jobs. But viruses evolve quickly and, every so often, some part of the protein that composes an infectious virus's capsid (the shell that protects the viral genome) acquires just the right electrochemical properties for binding to a cell-surface protein, tricking the cell into rolling out a welcome mat for its unwelcome guest.
Coronaviruses are a little different. Their attachment-enabling proteins are the spiky ones protruding from their greasy envelopes, not their capsids. Benign coronavirus variants, which like rhinoviruses can cause common colds, attach to cells in the upper respiratory tract. The viral variant that's driving today's pandemic is dangerous specifically because a recent mutation in its envelope-associated spike protein has enabled it to latch onto cells in the lower respiratory tract -- lung and bronchial cells.
Viral mutation rates are much higher than bacterial rates, which are higher than those of our germ cells. And RNA viruses, including the coronavirus, mutate even more easily than DNA viruses do: Their polymerases (genome copying enzymes, discussed in part one) are typically less precise than those of DNA viruses, and RNA itself is inherently less stable than DNA.
So viruses, and particularly RNA viruses, easily develop resistance to immune surveillance as well as to standard antiviral approaches.
"Viruses are so mutation-prone that you need two or three antivirals at once to defeat their escape," as in the case of HIV therapy, Carette said.
The experimental antiviral drug remdesivir, now being fast-tracked in clinical trials at Stanford and elsewhere for COVID-19, targets RNA viruses' polymerases, shutting down viral replication. Because all viruses carry and require one or another version of these enzymes, the hope is that remdesivir, initially developed (not very successfully) for treating Ebola virus infection, may prove effective against the novel coronavirus strain propelling the present pandemic.
Viruses don't always kill the cells they take hostage. Instead, some become chronic. So-called retroviruses, such as HIV, sew their genes into the genome of the cells they've invaded. These silent genes can activate when our immune response is weakened, setting off new rounds of viral replication.
Those insertions add up. Viral DNA sequences make up fully 8% of our genome -- in contrast with the mere 1% that codes for the proteins of which we're largely made and that do most of the making.
"Our genome has been 'invaded' by previous encounters with retroviruses after infection of germline cells," Carette told me. "Through evolution, these retroviruses' genes have become inactive."
As always, there's an exception, said Carette: "An ancient viral gene has been repurposed to play an essential role in embryogenesis," the process by which an embryo forms and develops. The protein this gene encodes enables the fusion of two kinds of cells in the developing fetus's placenta, allowing nutrient and waste exchange between the developing embryo and the maternal blood supply.
Without them, that is, there'd be no us.
Image of the novel coronavirus from the Centers for Disease Control and Prevention